

A statistical platform for clinical and efficacy trial analysis, designed for researchers who need rigorous methods without the complexity.






Clinical and efficacy trial data analysis is often fragmented: statisticians run scripts in isolation, researchers struggle to interpret outputs, and results are difficult to reproduce across projects. There was a clear need for a unified platform that could bring advanced statistical methods into an accessible, interactive environment without sacrificing scientific rigor.
TrialLytics was built to serve two audiences simultaneously: the statistician who needs full methodological control, and the clinical researcher who needs to explore results without writing code. The platform centralizes the most commonly used methods in efficacy trials into a single, configurable interface.
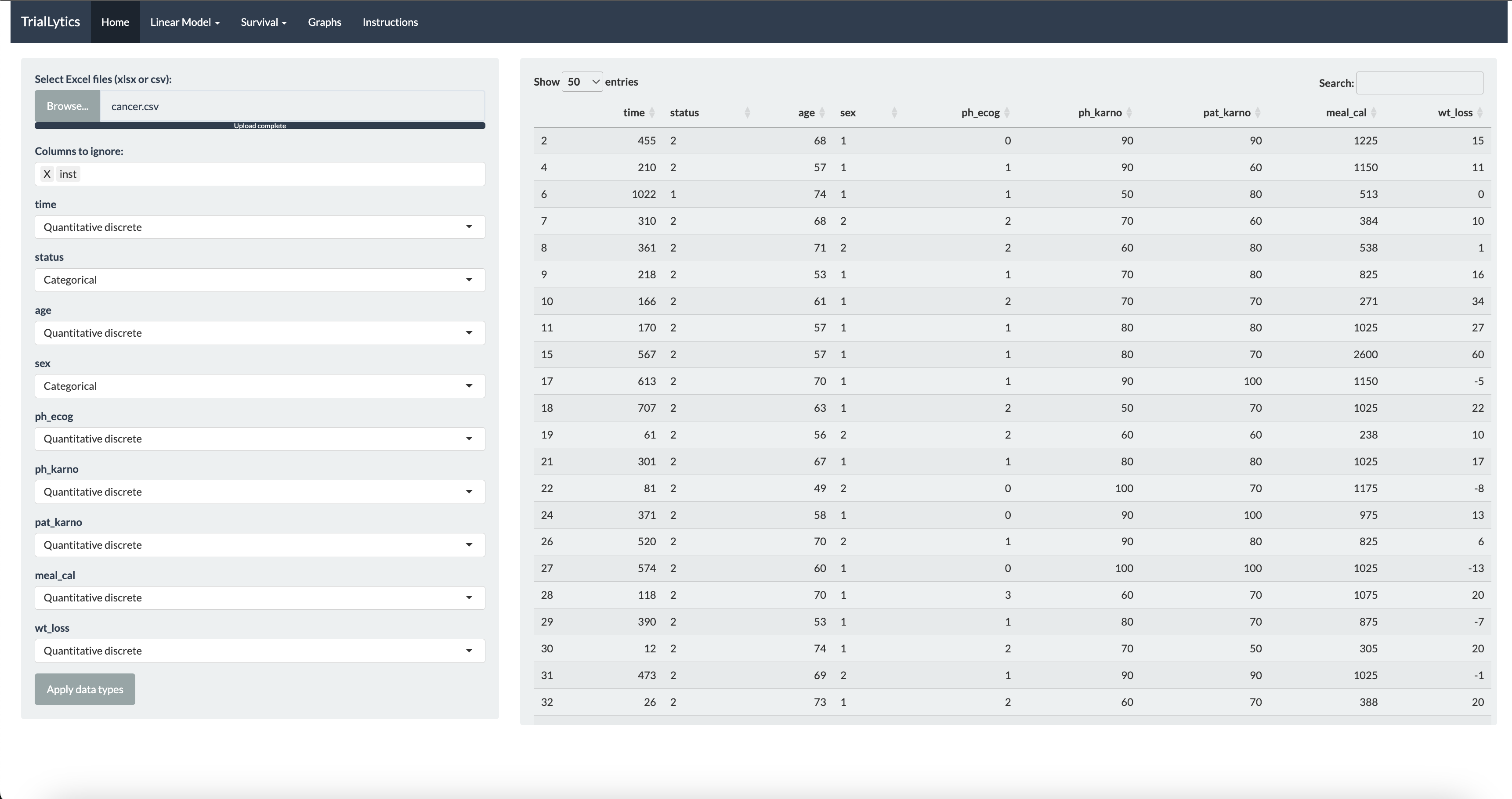
TrialLytics is an interactive web application that covers the full statistical workflow of a clinical or cosmetic efficacy trial. From data upload to formatted outputs, researchers can run analyses, interpret results, and export reports directly from the platform.
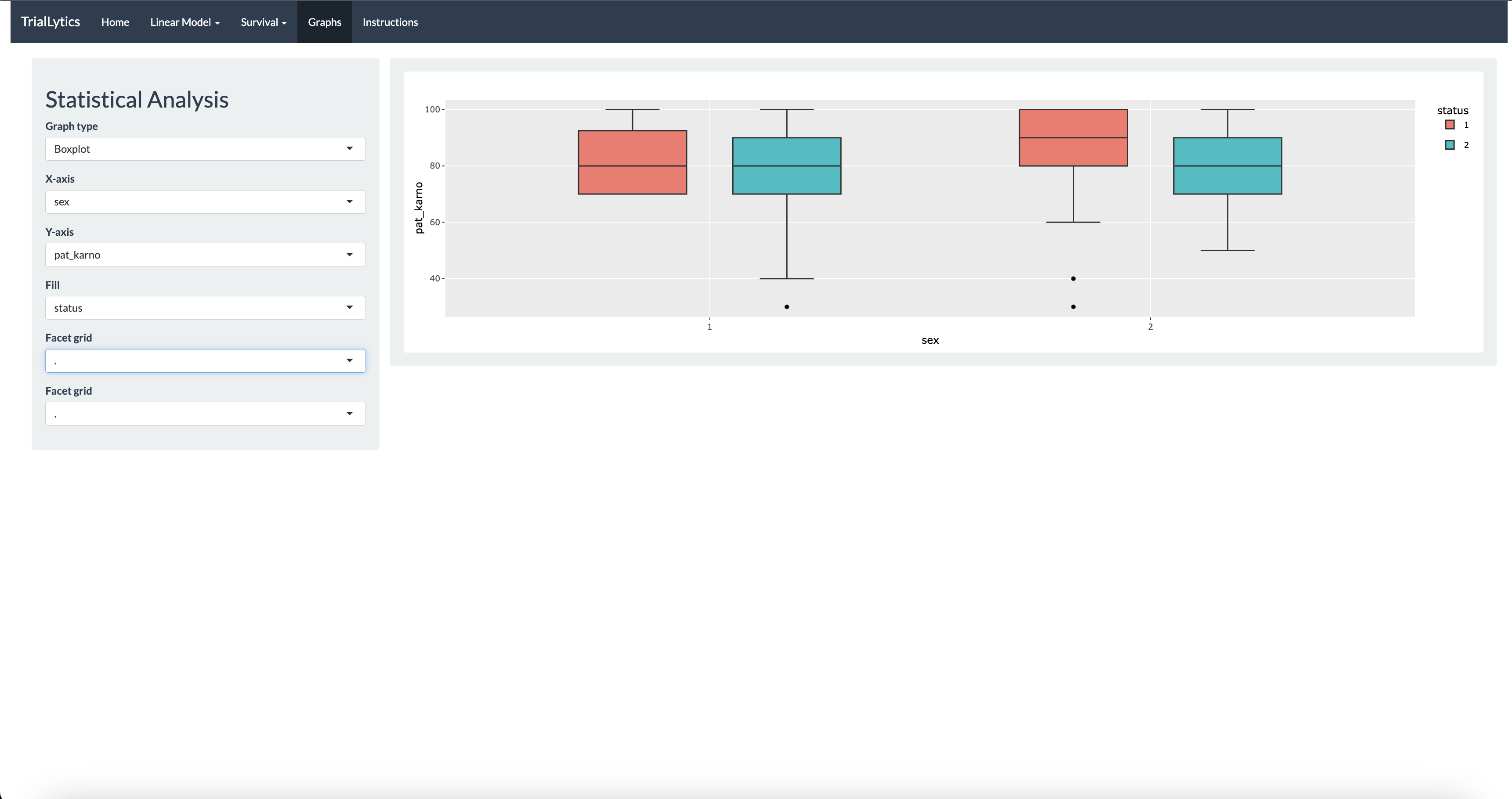
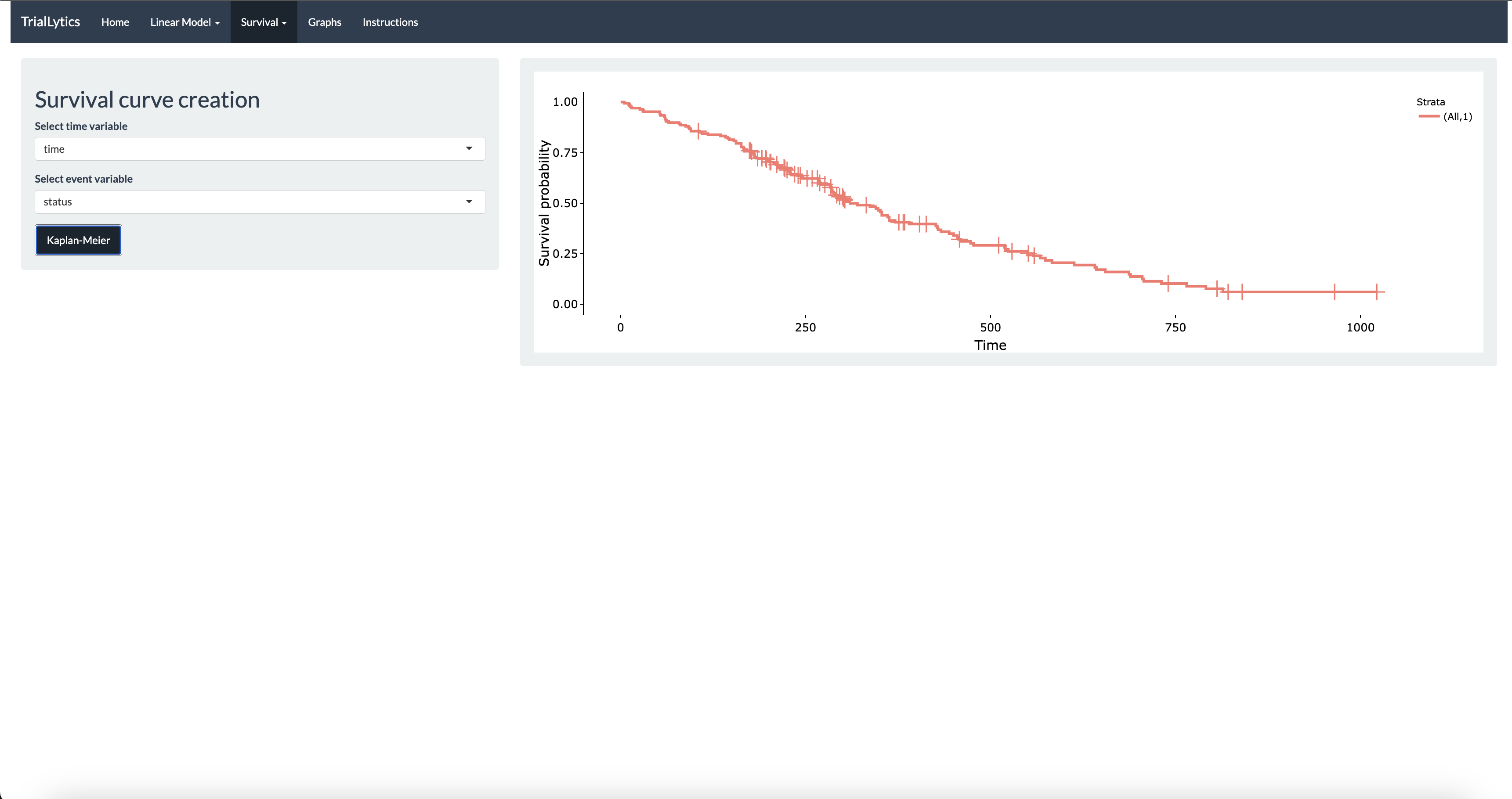
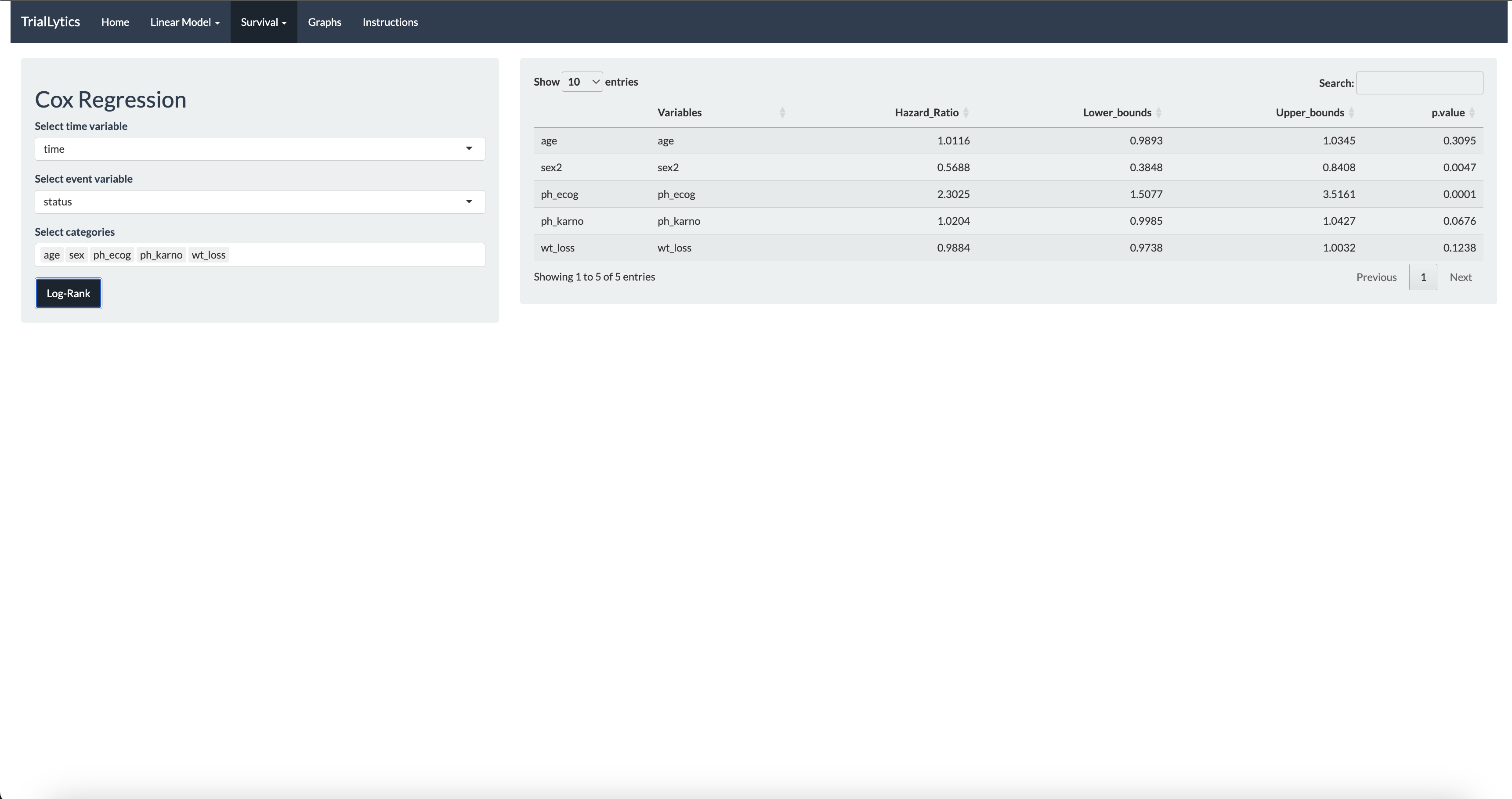
The platform supports mixed-effects models for longitudinal and repeated-measures data, Cox regression with hazard ratio interpretation, ANOVA and linear models for multi-group comparisons, and survival analysis with Kaplan-Meier curves. All visualizations are powered by Plotly, enabling interactive exploration of results.
Key outcome
Non-statistician researchers can independently run advanced analyses and interpret results, significantly reducing the bottleneck on data science resources within R&D teams.
TrialLytics is applicable across several R&D contexts:
Statistics
PK/NCA and bioequivalence platform with a full pipeline: ingestion, estimation, diagnostics, and reporting.
Data Engineering
Automated CDISC SDTM/ADaM pipeline orchestrated with Dagster for reproducible clinical data processing.
AI / ML
Graph model to suggest compatible substitutes for cosmetic formulations.